Abstract
Chronic wounds, particularly those associated with diabetes, pose a significant clinical challenge due to their impaired healing dynamics and lack of reliable and standardized preclinical models. This pilot study aimed to establish a diabetogenic, immunocompetent, hairless mouse model (SKH1 strain) to simulate prolonged wound healing. Diabetes was induced by streptozotocin administration, followed by the creation of full-thickness dorsal skin wounds. Wounds were treated with either saline or nanofibrillated cellulose hydrogel as a model treatment. Wound healing progression and blood glucose were monitored, and histopathological assessments were performed after a 14-day experiment. In addition, for the first time, the Thermidas thermal imaging system was used in an in vivo mouse model to evaluate skin temperature. Results demonstrated that diabetes induction successfully prolonged wound closure by 5 days compared with the previously described acute wound model in the same strain with the identical protocol without streptozotocin (STZ) induction. Histopathological analyses showed increased macrophage activity (16.2% vs. 2.2% in the treatment groups and 10.2% vs. 0.3% in the control groups) and decreased collagen deposition (12.2% vs. 43.2% in the treatment groups and 17.6% vs. 27.4% in the control groups), suggesting prolonged wound healing. These findings support the use of hairless SKH1 mice as a viable model for studying prolonged diabetic wound healing and evaluating future therapeutic candidates.
Graphical Abstract
Impact statement
Chronic wounds are often referred to as a silent epidemic and represent a growing global challenge with increasing prevalence each year. The development of novel bioactive wound therapies is hindered by the lack of standardized pre-clinical models and limited understanding of prolonged wound-healing pathophysiology. In this study, together with our previous work, we provide new insights into the potential of the SKH1 nude mouse strain as a versatile model for both acute and chronic wound healing research. To our knowledge, this is the first methodological study directly comparing diabetogenic and healthy SKH1 mice in a full-thickness wound model, revealing important differences in wound-healing pathology and treatment candidate efficacy. Publication of this work offers the opportunity to advance more standardized, reproducible wound-healing models that can accelerate therapeutic development while adhering to the principles of the 3Rs.
Introduction
An aging population and the rising prevalence of chronic diseases, such as diabetes and cardiovascular disorders, present global challenges in managing chronic wounds. Chronic wounds have been reported to cost up to 97 billion USD annually [] and increase the risk of mortality, particularly in patients with diabetic foot ulcers []. Insufficient homecare and the development of antibiotic resistance have led to a vicious cycle of chronic wound treatment [], which will not be resolved without novel treatments and a better understanding of wound pathophysiology.
Diabetes is a leading systemic contributor to chronic wound development, primarily by inducing local ischemia, elevating protease activity, and impairing skin cell function due to persistent hyperglycemia [, ]. While the current standard of care (including wound cleansing, pressure offloading, and infection control) remains essential, alternative therapies are being explored to enhance oxygen delivery, stimulate angiogenesis, and restore cellular activity. These include negative pressure wound therapy, bioactive hydrogels, and the application of growth factors [, ]. However, despite their promise, these approaches have yielded inconsistent results and lack standardized protocols, limiting their integration into routine chronic wound management.
The development of effective wound therapies is hindered by the heterogeneity of chronic wounds and the complexity of the wound healing process, making it difficult to establish standardized and reproducible preclinical models. Acute and chronic wounds differ significantly in key biological processes, further complicating both in vitro and in vivo modeling. The classification of wounds as acute or chronic is primarily determined by their healing duration, which itself depends heavily on the wound’s original size and location []. A wound is considered chronic if it fails to progress through the four partially overlapping healing phases (hemostasis, inflammation, proliferation, and maturation) within approximately 12 weeks. Beyond delayed or incomplete closure, chronic wounds are also characterized by persistent inflammation and impaired cellular function, often influenced by local or systemic conditions such as chronic diseases or infections []. In diabetes, for instance, angiogenesis—the formation of new blood vessels from existing vasculature—is impaired, resulting in insufficient oxygen and nutrient delivery to the wound site and contributing to delayed healing []. This vascular deficiency disrupts the balance of growth factors and cytokines, impairs epithelialization, and delays wound closure [, ]. Elevated levels of reactive oxygen species and altered macrophage activity lead to prolonged inflammation, as macrophages in chronic wounds fail to undergo the normal transition from pro-inflammatory (M1) to pro-regenerative states (M2) []. This sustained inflammation negatively impacts the formation of fibrous tissue and angiogenic activity []. Additionally, later stages of healing, including proliferation and maturation, are disrupted. Increased metalloproteinase activity and altered fibroblast function lead to an imbalance in collagen production [, ]. Such disturbances can result in excessive or pathological scarring, for which no definitive treatment currently exists.
Although research has focused on reducing the use of in vivo models with versatile three-dimensional in vitro models, the use of animals remains the gold standard in medical device and medicinal drug development for wound treatment. From the anatomical perspective, porcine models provide the most similar skin structure to human skin when comparing skin thickness, layers, and hair density []. Conversely, a more standardized genetic background and established genetic modifications, lower cost, and simpler habitat maintenance mean that mouse models are the most common in vivo model. However, this causes difficulties when translating in vivo findings to the clinic due to the larger differences between mice and humans, such as differences in skin thickness and contraction and its attachment to underlying tissue []. The most common method to study diabetic wounds in vivo is with streptozotocin (STZ)-induced mice []. STZ is a chemotherapeutic agent for the treatment of pancreatic -cell carcinoma, the cells in which normal insulin production occurs. In mice, STZ causes necrosis of the -cells, decreasing insulin production as in type I diabetes []. Although STZ induction is widely used, the protocols differ substantially between laboratories in terms of the experimental setups, e.g., variations in STZ dosing protocol, animal strain, experimental endpoints, animal diet, and data monitoring. Additional challenges come from the lack of both acute and prolonged wound healing models in the same experimental modality, challenging the understanding of the pathological differences.
Although most studies using STZ induction in rodents are considered disease pathology models, the model has also been used in chronic wound healing research. However, most models are generated with strains with typical skin and hair, which decreases the similarity to humans []. This challenge can be addressed by using hairless mouse strains, where the lack of hair and hair-follicle–derived wound-healing stem cells may offer a more suitable foundation for developing an in vivo wound healing model [, ]. Although these strains retain certain biological limitations, like differences in skin structure [] and the wound closure process [, ], they allow for easier wound creation, more consistent monitoring of the healing process, and improved capacity for thermographic assessment compared with other strains. Currently, there are few chronic wound models with immunocompetent hairless mouse strains, which encouraged us to generate a prolonged wound healing model in hairless SKH1 mice using a standardized STZ induction protocol. In addition, our previous study using SKH1 mice to generate an acute full-thickness wound model with an identical protocol to evaluate the efficacy and safety of wound treatment candidates was successful; it was carried out with good animal welfare, simplified wound creation, good monitoring of wound healing, and evaluation of possible changes in the skin during the experiment.
In this pilot study, our aim was to produce a tractable prolonged full-thickness wound healing model using STZ-induced hairless SKH1 mice, which has been used previously in our acute wound model study without diabetes induction []. For brevity, data in the prolonged wound healing model are henceforth referred to as “chronic wounds”. Based on this study we emphasize the following: 1) the potential for future standardization of current in vivo wound models by using the same strain and experimental set up in both acute and prolonged wound healing experiments; 2) the use of non-invasive monitoring tools to evaluate the progression of wound healing and possible infection with thermograph analysis; and 3) the use of hairless mice for simplified wound monitoring, and avoidance of ethically contentious methods such as mechanical prevention of wound closure. Through comparison with data from the previous acute wound model, changes in wound healing physiology, including epithelialization, inflammation, angiogenesis, and collagen deposition, could be observed in response to the same treatments. We hypothesize that hyperglycemic SKH1 mice with prolonged wound healing characteristics, including longer wound closure time, induced macrophage activity, and reduced collagen deposition, can be produced using STZ induction. By considering both acute and prolonged wounds, this study indicates the importance of standardized preclinical models for future wound care development.
Materials and methods
Model treatment formulations
To illustrate the effects of the material candidate in the chronic wound model, medical-grade nanofibrillated cellulose (NFC) hydrogel (FibGel, UPM Biomedicals) [2.9% (m/v)] was used as a model treatment as in our previous publication []. NFC hydrogel was diluted into 0.8% (m/v) with sterile water before use. 0.9% (m/v) NaCl was used as a control treatment.
Streptozotocin injection
Streptozotocin (STZ) (Sigma-Aldrich, batch 0661505.4, MO, USA) was solubilized in NaCl 0.9% (batch 214328131, Braun) into a final concentration of 8.0 mg/mL. STZ was prepared fresh daily and injected intraperitonially within 15 min of preparation. STZ bolus (40 mg/kg body weight) was administered daily for 5 consecutive days as previously described [] using a Myjector 27Gx1/2″ Terumo 0.3–0.5 mL insulin syringe. The dosing volume was 5 mL/kg. The injection site of skin was wiped with 70% EtOH prior to injection. Animals were not fasted before STZ injection to avoid additional stress factors for mice.
Animals
STZ induction was performed on 10 male SKH1 mice (Crl:SKH1-Hrhr, SPF, Charles River) at age 8–9 weeks. In total, nine of the STZ-induced mice underwent the chronic wound model. The pilot animal experiments were approved by the National Project Authorization Board of Finland (license number ESAVI-25539-2024), and the study conformed to the following guidelines: DIRECTIVE 2010/63/EU of the European Parliament and the Council, Finnish Act (497/2013), Government Decree on the Protection of Animals Used for Scientific or Educational Purposes (564/2013), and Guidance document on the Recognition, Assessment and Use of Clinical Signs as Humane endpoints for Experimental Animals Used in Safety Evaluation, Environmental Health and Safety Monograph Series on Testing and Assessment (No 19. OECD 2000). The reporting of this animal research followed the ARRIVE 2.0 (Animal Research: Reporting In Vivo Experiments) guidelines [] in the spirit of the pilot study.
This non-Good Laboratory Practices (GLP) animal experiment was performed in a GLP-certified Central Animal Laboratory, Turku University, Finland. The acclimatization period before the first experiment procedure was 6–13 days. The first STZ injection was given after the acclimatization period, and injections were given on 5 consecutive days. The mice were housed four to five animals per cage before experimentation and individually during the experiments, starting on study day 0. Cellulose paper and cardboard houses were used as environmental enrichment. Laboratory room temperature was 21 °C ± 3 °C, relative humidity was between 40% and 60%, and artificial lighting followed a 12-hour light, 12-hour dark cycle. A laboratory rodent chow diet (Teklad2920, Inotiv) and water were offered ad libitum, and the animals were cared for according to the standard operating procedures of Central Animal Laboratory. The clinical status was checked twice daily during the experiment period. The animals were weighed on study days 0, 2, 5, 6, 8, 10, 12, and 14 after wound surgery without dressing.
Blood glucose monitoring
The blood glucose of the mice was measured for the first time after the first STZ injection using the ACCU Check Aviva glucose meter and Accu-Chek Aviva Blood Glucose Test Strips (lot 690720). The validity of the meter was checked by using Accu-Chek Guide control solutions (lot 24700675). Blood for glucose determination was taken from the tail vein by needle prick using a 27G needle and analyzed with a glucometer (0.6 µL of blood). When the blood glucose level was ≥15 mmol/L (corresponding to 270 mg/dL) in two consecutive measurements, a mouse was diagnosed with diabetes.
When blood glucose rose to ≥20 mmol/L, blood glucose was monitored daily, and insulin treatment was started at ≥25 mmol/L. The insulin doses were administered to mice once or twice a day, depending on the glucose level response (Lantus 100 IU/mL, 0.5 IU accuracy, batch 3F231A, insulin pen JuniorSTAR). The starting dose was 0.5 IU per mouse per day, after which the dose varied between 0.5 IU and 3.0 IU (Table 1).
TABLE 1
| Glucose level | Insulin treatment/day |
|---|---|
| <20 mmol/L | No insulin |
| 20–25 mmol/L | 0.5 IU |
| 25–30 mmol/L | 0.5–2 IU |
| >30 mmol/L | 1–3 IU |
Glucose levels and the used insulin treatment dose.
Surgical procedure
Full-thickness wounds were created as described previously []. Briefly, 22 days after the last STZ injection, mice were preoperatively given a subcutaneous injection of buprenorphine (Bupaq multidose vet 0.1 mg/kg, Ritchter Pharma) and carprofen (Rimadyl vet 16 mg/kg, Zoetis) and anesthetized before the surgical procedure with isoflurane (3.5% Attane Vet 1,000 mg/g). Prior to incision, infiltrative local anesthesia of lidocaine (Lidocaine 4 mg/kg, Baxter) was applied under the skin. Rimadyl was also administered 6–8 h after surgery and every 10–13 h on study days 1 and 2 as a postoperative treatment. Surgical sites were disinfected with a skin disinfectant, and incisions were made on both sides of the animal’s back with sterile scissors and tweezers, first to the right side and then to the left side. The veterinarian was blinded from the dosing sites of the treatment. The wounds were measured, and photographs were taken immediately after surgery. After applying the model treatments on the right side and saline as a control on the left side, wounds were covered with transparent, non-occlusive polyurethane film.
A total of nine mice underwent surgery since one animal was hypoglycemic on the operation day (blood glucose <1.0 mmol/L) and had a decreased level of consciousness. The animal was treated with 0.5 mL of 10% sugar-water solution (D-sucrose, p.o.) and honey on the mouth mucous membranes. After 1 h, the animal was given 0.2 mL of 50% sugar solution. After 50 min of the last sugar application, the blood glucose level was 4.7 mmol/L. Subsequently, 10% sugar solution was provided ad libitum. After 3 h, the blood glucose level was 24.0 mmol/L. Blood sugar levels and insulin treatment were later performed in the same way as with animals that underwent surgery for welfare purposes, but are not plotted in the figures.
Wound monitoring
The length and width of the wounds were measured immediately after their creation and on study days 2, 5, 6, 8, 10, 12, and 14 using our previously published protocol with a calibrated digital caliper (Mitutoyo 0–150 mm) []. The analyst measured the wounds as a one-time method to keep the objectivity of the wound measurements. The wound area was calculated with the following equation according to Moreira et al. []:
At the end of the study, mice were weighed, all macroscopic abnormalities at the wound site were recorded, and photographs of the wounds were taken. Blood samples (ca. 600 μL) were taken by heart puncture under isoflurane anesthesia in K2E Microtainer tubes (Becton Dickinson, United States, NJ, lot 4174515) for hematological analyses, which were implemented as blinded with sample coding without identifying the wound. Hematology test analyses were performed in EDTA blood tubes according to the protocol of Central Animal Laboratory using VetScan HM5 hematological analyses (Abaxis, United States). A piece of skin (1 cm2), including the wound site, was removed and placed into 10% phosphate buffered neutral formalin (Oy Reagena Ltd, Finland, lot CB19/1). Subsequently, tissue samples were embedded in paraffin and cut into 4 µm sections for further histopathological analyses.
Thermal monitoring of the wounds
For wound temperature monitoring, a Thermidas IRT-384 Tablet (Vet VistaClinic, Software 1.4.2, Thermidas Oy, Tampere, Finland) was used. Images were taken in the same operating room (ambient temperature of 21–23 °C) within a 2-hour period on days 0, 2, 5, 6, 8, 10, 12, and 14 at the same time as the weight and wound measurements. Before imaging the mice, a blank image from the operating room table was taken to calibrate the temperature range. Images were taken approximately 15 cm from the animal. Animals were anesthetized before imaging, and the total operation time was approximately 4 min per animal. The thermal resolution of the images was 384 × 288 pixels. The temperature of the wounds was compared to healthy skin on the same animal.
Histopathology and immunohistochemistry
Wound tissue sections (4 µm thickness) were stained with hematoxylin and eosin (HE) for histological examination. A veterinary pathologist (J.L.) blinded to the sample identity assessed the staining. Masson’s trichrome (MT) staining was used to evaluate collagen deposition by calculating the blue color intensity of the wound area.
Immunostaining was performed as reported previously by Koivuniemi et al. [] and Koivunotko et al. []. Briefly, tissue sections were deparaffinized (3 2 min in xylene, 2 10 min in 100% ethanol, 2 10 min in 94% ethanol and 2 5 min in distilled H2O). Antigen retrieval was carried out in 10 mM citrate buffer (Merck, Germany) with 0.05% Tween 20 solution (Merck) (pH 6) at 99 °C for 3 × 10 min. After antigen retrieval, endogenous peroxidase activity was blocked with 3% H2O2 (Merck) for 10 min. Sections were then blocked for 1 h in 5% bovine serum albumin (BSA, Merck) in Tris-buffered saline with Tween 20 (TBS-T, Merck). Next, sections were blocked with endogenous biotin (Avidin/Biotin blocking kit, Vector Laboratories, CA, USA) for 15 min with Avidin first and then 15 min with Biotin.
Tissue sections were incubated overnight at 4 °C with anti-rabbit CD31/platelet endothelial cell adhesion molecule (CD31/PECAM-1, 1:100, CAT: NB100–2284, Novus Biologicals, UK) or 1 h at room temperature with anti-rabbit ionized calcium-binding adaptor molecule 1 (Iba-1, 1:500, CAT: 019-19741, FujiFilm, USA) or anti-rabbit lysozyme (LZM, 1:1000, CAT: A0099, Agilent, CA, USA) as a primary antibody in 3% BSA/TBS-T. After washing with TBS-T, tissue sections were stained for 1 h at room temperature with goat anti-rabbit IgG as biotinylated secondary antibody (1:1000 for CD31 and 1:200 for Iba1 and LZM, Abcam, UK) in 3% BSA/TBS-T. VECTASTAIN Elite ABC reagents (Vector Laboratories) were used for antibody detection by staining the tissue sections for 30 min at room temperature, after which they were treated with 3,3′-diaminobenzidine (DAB) HRP substrate treatment (Vector Laboratories). All stained tissue sections were counterstained with hematoxylin, dehydrated, and covered with cover clips using a mounting medium (Coverquick 2000, VWR International, PA, USA).
Histoscanning was performed with a Pannoramic 250 Flash III brightfield digital slide scanner (3DHISTECH Ltd., Hungary) at the Histoscanner core facility (University of Helsinki) using the updated coding from the original one to increase the blindness of the image analyses. The thickness and length of the neo-epithelium and dermis were measured with CaseViewer (3DHISTECH Ltd., version 2.4). The intensity of the stained fibrous connective tissue and macrophage infiltration was evaluated using object and pixel classifications in QuPath 0.4.3 software []. To compare the results from the prolonged wound healing model with the previously described acute wound model using the same histopathological staining protocol [], acute wound tissue section analyses were repeated with the same threshold settings. Thresholds in different analyses were as follows: 0.4 for iba-1 and LZM staining, 0.15 for CD-31 staining, 0.64 for MT staining.
Statistical analyses
The data are presented as mean standard deviation (STDEV). For the normally distributed data, statistical significance was determined with an unpaired t-test for two variables and a one-way ANOVA and Tukey HSD post hoc test for others. To evaluate the effects of glucose levels [defined as low (<23.4 mmol/L) and high (>23.4 mmol/L)] on the results, multivariate analyses were performed with a Wilks’ Lambda test. Significance was concluded when *p < 0.05, **p < 0.01, ***p < 0.001, or ****p < 0.0001.
Results
Diabetes was successfully induced in all 10 animals
Diabetes was induced in all animals (blood glucose level within 21 days after the first STZ bolus injection. In this study, animals were not fasted before STZ injections or before blood glucose measurements. The blood glucose measurements were performed daily or twice daily to ensure animal welfare and titrate the insulin dose for each animal. The mean blood glucose level on the last experimental day was 27 mmol/L (STDEV 4.1 mmol/L), which was significantly increased (p < 0.001) compared to the 16 days prior to surgery (Figure 1A). Based on clinical monitoring and weight (Figure 1B), even animals with high blood glucose levels (>30 mmol/L) were in good condition during the whole experiment. The non-significant weight decrease (3–9%) in some animals was correlated with higher blood glucose (>23.4 mmol/L) but was insignificant in multivariate analyses (p = 0.085).
FIGURE 1
Hematological analyses showed increased values in most of the measured parameters (Table 2) when compared with the acute wound model []. In the case of hematocrit, mean corpuscular hemoglobin, plateletcrit, and the total concentration of monocytes and neutrophils, no changes (less than 0.5-unit difference) were observed between the models. On the other hand, mean corpuscular volume, red blood cell distribution, and percentage of lymphocytes were decreased. However, no clinically relevant differences were observed.
TABLE 2
| Parameter | Unit | Value, mean (STDEV n = 9) | Previous acute wound model value (n = 3–6) | Reference value [–] |
|---|---|---|---|---|
| WBC- White blood cell | 109/L | 5.6 (2.5) | 4.1 (1.5) | 9.3 (1.8) |
| RBC- Red Blood Cell | 1012/L | 11.1 (0.3) | 9.9 (2.1) | 9.4 (0.4) |
| HGB- Hemoglobin | g/L | 157.6 (7.5) | 142 (186.7) | 120 (10.2) |
| HCT- Hematocrit | % | 52.4 (4.0) | 52.9 (4.1) | 49.4 (2.2) |
| MCV- Mean Corpuscular Volume | fl | 47.2 (3.0) | 53.7 (2.3) | 53.9 (2.4) |
| MCH- Mean Corpuscular Hemoglobin | pg | 14.2 (0.4) | 14.4 (0.6) | 17.1 (0.4) |
| MCHC- Mean Corpuscular Hemoglobin Concentration | g/L | 301.9 (15.5) | 268 (16.6) | 317.4 (9.8) |
| PLT- Platelet | 109/L | 431.2 (95.8) | 335 (36.5) | 285–890 |
| PCT- Plateletcrit | % | 0.3 (0.1) | 0.2 (0.02) | N/Av |
| MPV- Mean Platelet Volume | fl | 6.6 (0.4) | 6.1 (0.9) | 4.6 (0.1) |
| PDWs- Platelet Distribution Width | fl | 7.4 (0.4) | 6.4 (1.8) | 7.7 (1.1) |
| PDWc- Platelet Distribution Width | % | 28.4 (0.7) | 26.6 (3.2) | N/Av |
| RDWs- Red blood cell Distribution | fl | 32.7 (1.7) | 38.3 (1.2) | 29.1 (3.3) |
| RDWc- Red blood cell Distribution | % | 21.6 (0.5) | 19.9 (0.8) | 16.4 (0.4) |
| LYM- Lymphocytes | 109/L | 4.1 (2.1) | 3.6 (1.7) | 5.7 (1.3) |
| MON- Monocytes | 109/L | 0.2 (0.1) | 0.2 (0.1) | 0.6 (0.2) |
| NE- Neutrophils | 109/L | 1.3 (0.6) | 1.0 (0.1) | N/Av |
| LY%- Lymphocytes | % | 71.9 (7.3) | 73.8 (3.4) | 61.5 (6.9) |
| MO%- Monocytes | % | 4.8 (2.2) | 4.9 (2.5) | 6.1 (1.3) |
| NE%- Neutrophils | % | 23.3 (6.0) | 21.3 (10.2) | N/Av |
| EOS- Eosinophils EO%- Eosinophils BAS- Basophils BA%- Basophils | 109/L % 109/L % | 0.0 | 0.0 | N/Av 0.6 (0.2) 0.0 (0.0) 0.0 (0.0) |
Hematological analyses of total blood samples after a prolonged wound healing model experiment. The results from the previous acute wound model in SKH1 [] with informed reference values are presented on the right-hand side of the table.
Wounds were closed by day 14
The endpoint of the experiment was on day 14, during which the first closed wounds were observed (<5 mm2) (Equation 1). No significant differences in wound closure, epithelialization, or fibrous tissue thickness between model treatment and control groups were observed (Figures 2A–C). The mean epithelium length in tissue samples was 1271 µm in treated wounds and 1207 µm in controls. The mean fibrous tissue thickness measured from the dermis site was 385 µm in treated wounds and 348 µm in control wounds. Histological evaluation of the HE-stained sections by the pathologist (J.L) showed regrown epidermis to cover the wound areas in all mice, often exhibiting minimal to mild epidermal hyperplasia. Model treatment was present extracellularly as large homogenous translucent depots occupying over half of the wound area in most treated samples.
FIGURE 2
The wound area treated with model treatment showed significantly decreased temperature during the first week compared with healthy skin temperatures (8 °C lower temperature on day 0, 2 °C on day 6, Figure 2D; Supplementary Material 1). In addition, treated wounds were 3 °C colder compared with control wounds on day 0.
Blood vessel formation at the wound site
The presence of newly formed blood vessels was measured from CD31 stained histopathological samples. The percentual contrast area of the CD31 staining in wounds was >5% in all wounds (Figure 3). In addition, the analyses were also performed for previous histopathological samples from the acute wound model on day 9 [], but no significant differences were observed between CD31 stained tissue samples. In wounds with model treatment, standard deviation was higher than in control groups.
FIGURE 3
Increased infiltration of macrophages with higher activity in prolonged wound healing model
Macrophage cell infiltration was evaluated with both total macrophage staining (Iba-1) and active macrophage staining (LZM) (Figures 4A–D). The contrast analyses were also performed for previously obtained Iba-1 and LZM stained histopathological samples from the acute wound model on experimental day 9 [
FIGURE 4
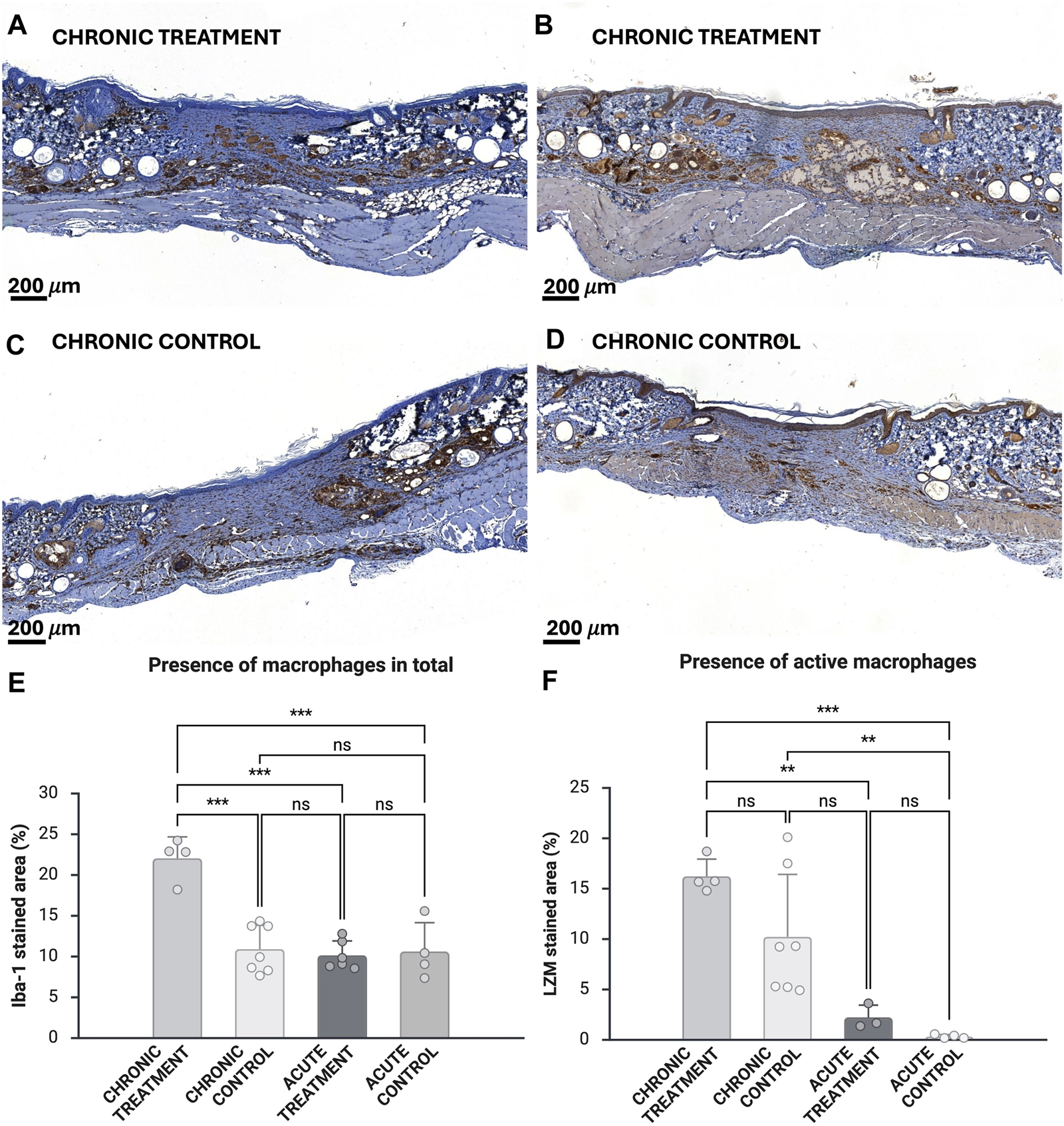
The presence of macrophages in total stained with Iba-1 (A,C) and active macrophages stained with LZM (B,D). Upper tissue sample images are from treated chronic wounds and lower ones from chronic control wounds. Percentual area of the iba-1 stained macrophages (n = 4 for chronic treatment, n = 6 for chronic control, n = 6 for acute treatment, n = 4 for acute control) (E) and LZM stained macrophages (n = 4 for chronic treatment, n = 7 for chronic control) (F). Analyses were also performed for previously obtained histopathological samples (n = 3 for acute treatment, n = 4 for acute control) [
In the histological examination the treated wounds showed a stereotyped moderate inflammatory reaction, consisting of a thin rim of macrophages surrounding and infiltrating the hydrogel depots and single macrophages that contained modest amount of foamy hydrogel material as well as a moderate macrophage and lymphocyte infiltrate outside of the depots. Very few multinucleated giant cells were present. In comparison, most control wounds displayed mild to moderate mononuclear inflammatory cell infiltrate in the wound area.
Notably, most samples, regardless of treatment, exhibited pronounced, focally extensive hypodermal and deep dermal pyogranulomatous inflammation affecting damaged hair follicles and sebaceous glands (Supplementary Material 2). Granuloma formation was most prevalent at the wound borders and displayed a striking increase compared to the acute wound model [
Collagen deposition was slowed down in prolonged wound healing model
The blue color density of histopathological MT samples was measured to evaluate collagen deposition at wound sites (Figure 5). Based on the contrast analyses of chronic wounds on day 14 (Figures 5A,B) and acute wounds on day 9, collagen deposition was significantly lower (12.2 0.09% and 23.7 0.1%) in chronic wounds compared with treated acute wounds (43.2% 18.6%; p = 0.003 between treatment groups, p = 0.004 chronic control vs. acute treatment) (Figure 5C). Higher blood glucose levels (>23.4 mmol/L) correlated with lower collagen deposition (p < 0.01).
FIGURE 5
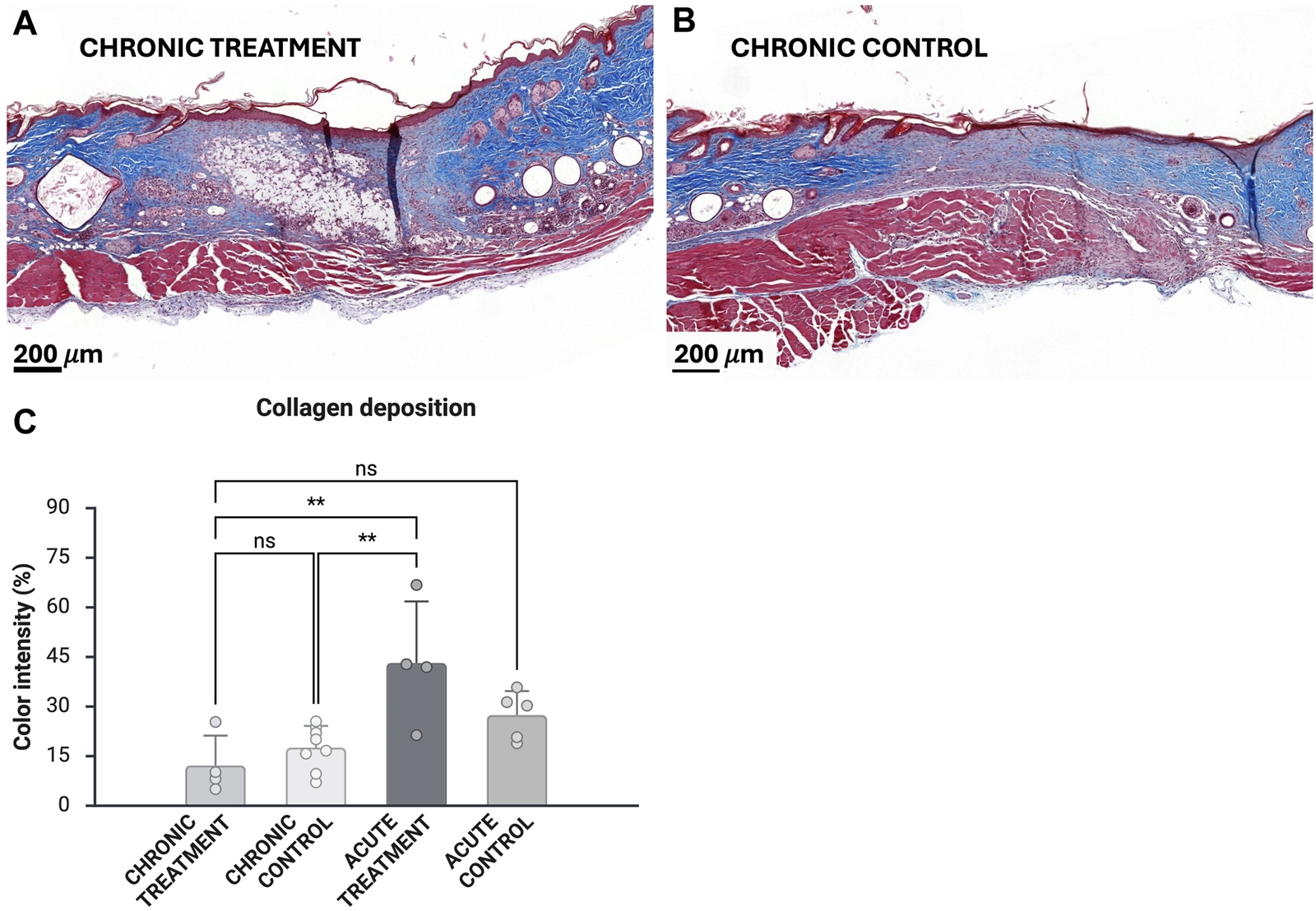
Masson trichrome analyses of treated chronic wounds (A) and control wounds (B). Collagen deposition was measured from blue staining intensity (n = 4 for chronic treatment, n = 7 for chronic control) (C). Analyses were also performed for previously obtained histopathological samples (n = 4 for acute treatment, n = 5 for acute control) [
Histological evaluation revealed plump horizontally orienting fibroblasts and palely staining collagen in the regenerating dermal connective tissue in both control and treated samples, suggesting maturing fibrous tissue with subjectively scarce collagen formation as well as sparse to moderate neovascularization (Supplementary Material 2). One control wound exhibited granulation tissue and one treated wound immature collagenous connective tissue.
Discussion
In this study, diabetes was successfully induced in all tested SKH1 mice. Furthermore, successfully prolonged full-thickness wound healing was observed in all surgical animals when compared to our previously described acute full-thickness wound model with the same surgical procedure and mouse strain [
FIGURE 6
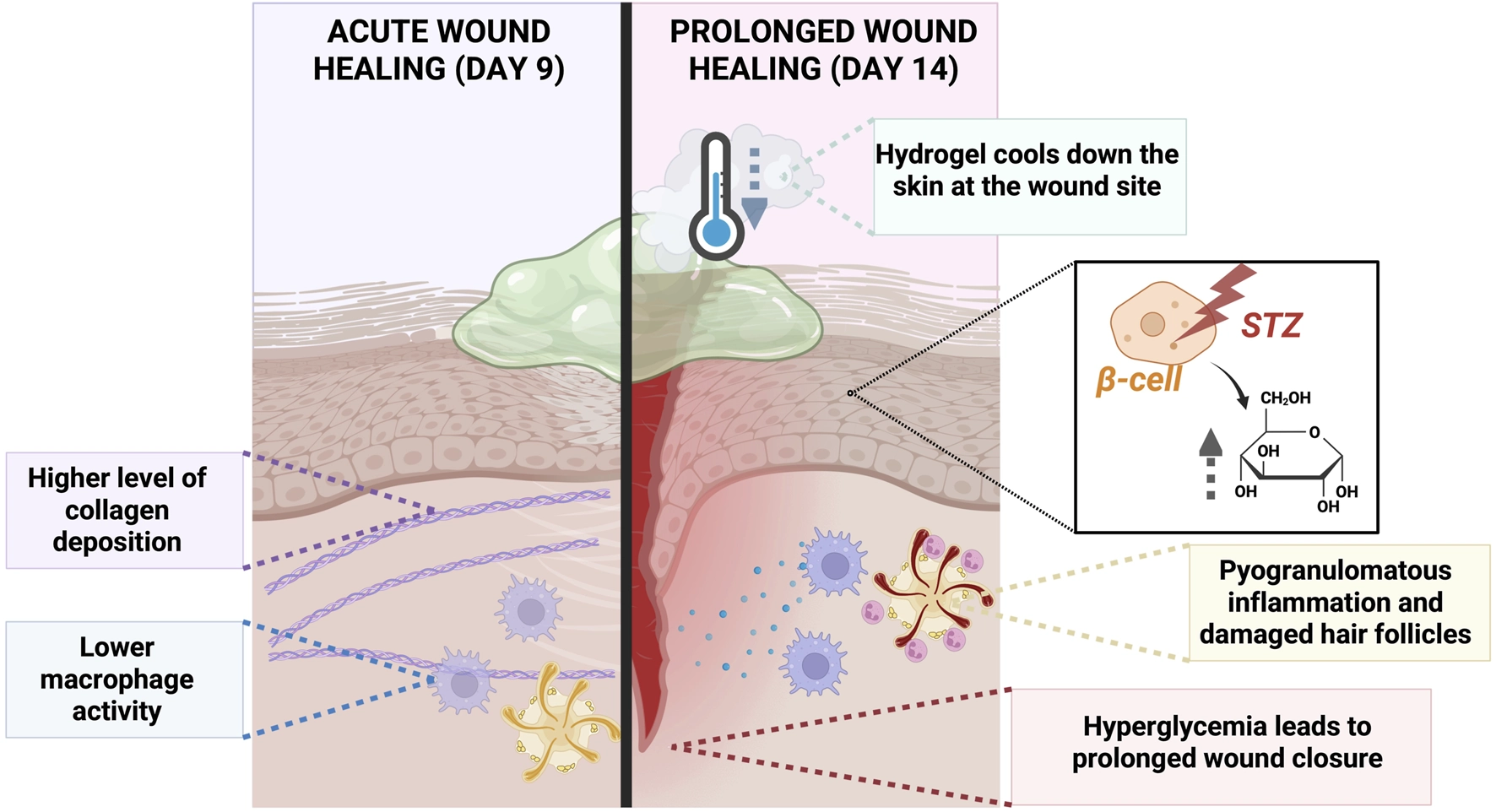
Graphical summary of the comparison between the acute wound model and prolonged wound healing model implemented in SKH1 mice.
In addition, animal welfare was closely monitored throughout the study by daily assessment of weight, behavior, and the risk of insulin dose-related hypoglycemia, all while maintaining on ad libitum diet. Since STZ induction has been reported to be equally diabetogenic to fed or fasted mice, fasting was not necessary in this model [
In addition to blood glucose level monitoring, hematological parameters were measured at the end of the experiment. Based on the hematological parameters, a slight increase in the number of white blood cells, red blood cells and platelets were observed. However, these changes were not significantly different from our previous data [
Higher blood glucose levels in SKH1 mice might have led to prolonged wound closure compared with similar size wounds in the acute wound healing model [
Thermidas thermal imaging system was used to measure temperature changes in differently treated wounds and healthy skin. In general, temperature monitoring can non-invasively detect early diagnosis of prolonged inflammation, the state of angiogenesis and the total progress of wound healing. In addition, thermal analyses indicate treatment candidate effects on wound site which may reflect on healing outcome [
A key issue in diabetic prolonged or chronic wounds is disturbed blood vessel formation due to the effects of high blood glucose concentration on the endothelial cell functions [
Another major factor in the progression of wound healing and activation of angiogenesis is the change in presence of pro-inflammatory macrophages into tissue regenerative ones, roughly categorized as M1 and M2 phenotypes [
The imbalance in macrophage activity in wound healing may lead to increased levels of reactive oxygen species damaging the ECM during re-modeling [
Conclusion
To our knowledge, no previous published work has compared acute wound healing and diabetic prolonged wound healing in hairless SKH1 mice. This pilot study may show the suitability of SKH1 mice for studying full-thickness wound healing under different conditions. Diabetes was induced in all mice using repeated STZ induction without complications. The markedly prolonged wound closure time, elevated macrophage activity, and reduced collagen deposition reflected key features of chronic wounds. However, establishing a fully representative chronic wound model requires additional considerations, like reduced oxygen availability, and the potential influence of infection; these factors were not examined in this study. With careful monitoring, SKH1 mice may be used in future wound healing studies as acute and prolonged wound healing models that are highly tractable for STZ-induced diabetes. The sample size of this proof-of-concept study was deliberately small and relies on wound healing in SKH1 mice in previously published data. Therefore, studies with a larger sample size, including healthy control groups and other treatment candidates, should be performed in future. By offering the opportunity to generate standardized and reproducible in vivo models, the development of wound treatment candidates can be enhanced while adhering to the 3Rs principles.
Statements
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The animal experiments were approved by the National Project Authorization Board of Finland (license number ESAVI-25539-2024) and the study conformed to the following guidelines: DIRECTIVE 2010/63/EU of the European Parliament and the Council, Finnish Act (497/2013), and Government Decree on the Protection of Animals Used for Scientific or Educational Purposes (564/2013), and Guidance document on the Recognition, Assessment and Use of Clinical Signs as Humane endpoints for Experimental Animals Used in Safety Evaluation, Environmental Health and Safety Monograph Series on Testing and Assessment (No 19. OECD 2000).
Author contributions
EK drafted the manuscript. EK, CP, and MM were responsible for validation. EK was responsible for visualization. EK, JM, and MM were responsible for methodology. EK, JM, JL, and MM conducted the investigation. EK, JL, and MM performed formal analysis. EK and JL were responsible for data curation. EK, CP, and MY conceptualized the experiments. RH and MY acquired funding and were responsible for supervision. MY was also responsible for resources and project administration. All authors contributed to the article and approved the submitted version.
Funding
The author(s) declared that financial support was received for this work and/or its publication. EK acknowledges the Post Docs in Companies program 2025 funded by Jenny and Antti Wihuri Foundation. JM acknowledges the Doctoral Programme in Materials Research and Nanosciences (University of Helsinki, Finland). CP acknowledges the Finnish Cultural Foundation (grant no. 00250755). RH and MY acknowledge the Academy of Finland, GeneCellNano flagship-project (grant no. 337430) and RH acknowledges the Finnish Cultural Foundation (grant no. 00220283).
Acknowledgments
The authors gratefully acknowledge the expert care provided by the personnel of Central Animal Laboratory, University of Turku. The authors also thank the Finnish Centre for Laboratory Animal Pathology (FCLAP) for histological sample processing and HE, MT, Iba-1, and LZM staining, as well as the Histoscanner core facility at the University of Helsinki for supporting histological analyses. Both FCLAP and the Histoscanner core facility are supported by HiLIFE and Biocenter Finland. The Figures were created with Biorender.com.
Conflict of interest
MM was employed by the company Made Consulting Ltd Oy. EK was part of the Postdocs in Companies project in collaboration with UPM Kymmene. UPM Kymmene was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
The remaining author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Generative AI statement
The author(s) declared that generative AI was not used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.ebm-journal.org/articles/10.3389/ebm.2026.10857/full#supplementary-material
References
1.
NussbaumSRCarterMJFifeCEDaVanzoJHaughtRNusgartMet alAn economic evaluation of the impact, cost, and medicare policy implications of chronic nonhealing wounds. Value in Health (2018) 21(1):27–32. 10.1016/j.jval.2017.07.007
2.
ArmstrongDGTanTWBoultonAJMBusSA. Diabetic foot ulcers: a review. JAMA (2023) 330:62–75. 10.1001/jama.2023.10578
3.
OrfaliRGhaffarSAlAjlanLPerveenSAl-TurkiEAmeenF. Diabetes-related lower limb wounds: antibiotic susceptibility pattern and biofilm formation. Saudi Pharm J (2024) 32(6):102069. 10.1016/j.jsps.2024.102069
4.
TobalemMLévigneDModarressiAAtashiFVillardFHinzBet alHyperglycemia interacts with ischemia in a synergistic way on wound repair and myofibroblast differentiation. Plast Reconstr Surg Glob Open (2015) 3(7):e471. 10.1097/GOX.0000000000000443
5.
DorfNMaciejczykM. Skin manifestations in diabetes—what is new?Front Med (2025) 12:1640144. 10.3389/fmed.2025.1640144
6.
BaltzisDEleftheriadouIVevesA. Pathogenesis and treatment of impaired wound healing in diabetes mellitus: new insights. Adv Ther (2014) 31:817–36. 10.1007/s12325-014-0140-x
7.
YadavJPSinghAKGrishinaMPathakPVermaAKumarVet alInsights into the mechanisms of diabetic wounds: pathophysiology, molecular targets, and treatment strategies through conventional and alternative therapies. Inflammopharmacology (2024) 32:149–228. 10.1007/s10787-023-01407-6
8.
MortonLMPhillipsTJ. Wound healing and treating wounds differential diagnosis and evaluation of chronic wounds. J Am Acad Dermatol (2016) 74:589–605. 10.1016/j.jaad.2015.08.068
9.
GuoSDiPietroLA. Factors affecting wound healing REVIEW. J Dent Res (2010) 89 (3): 219–229. 10.1177/0022034509359125
10.
PatelSSrivastavaSSinghMRSinghD. Mechanistic insight into diabetic wounds: pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomed Pharmacother (2019) 112:108615. 10.1016/j.biopha.2019.108615
11.
GalkowskaHWojewodzkaUOlszewskiWL. Chemokines, cytokines, and growth factors in keratinocytes and dermal endothelial cells in the margin of chronic diabetic foot ulcers. Wound Repair Regen (2006) 14(5):558–65. 10.1111/j.1743-6109.2006.00155.x
12.
LinardCBrachetMStrup-PerrotCL’hommeBBussonESquibanCet alAutologous bone marrow mesenchymal stem cells improve the quality and stability of vascularized flap surgery of irradiated skin in pigs. Stem Cells Transl Med (2018) 7(8):569–82. 10.1002/sctm.17-0267
13.
HuntMTorresMBachar-WikstromEWikstromJD. Cellular and molecular roles of reactive oxygen species in wound healing. Commun Biol (2024) 7:1534. 10.1038/s42003-024-07219-w
14.
HeskethMSahinKBWestZEMurrayRZ. Macrophage phenotypes regulate scar formation and chronic wound healing. Int J Mol Sci (2017) 18. 10.3390/ijms18071545
15.
HirotaAEbiharaTKusubataMKobayashiMKobayashiKKuwabaKet alCollagen of chronically inflamed skin is over-modified and upregulates secretion of matrix metalloproteinase 2 and matrix-degrading enzymes by endothelial cells and fibroblasts. J Invest Dermatol (2003) 121(6):1317–25. 10.1111/j.1523-1747.2003.12637.x
16.
ChiNZhengSClutterEWangR. Silk-CNT mediated fibroblast stimulation toward chronic wound repair. Recent Prog Mater (2019) 1 (4): 16. 10.21926/rpm.1904007
17.
AnsellDMHoldenKAHardmanMJ. Animal models of wound repair: are they cutting it?Exp Dermatol (2012) 21(8):581–5. 10.1111/j.1600-0625.2012.01540.x
18.
ZomerHDTrentinAG. Skin wound healing in humans and mice: challenges in translational research. J Dermatol Sci (2018) 90(1):3–12. 10.1016/j.jdermsci.2017.12.009
19.
DuYWangJFanWHuangRWangHLiuG. Preclinical study of diabetic foot ulcers: from pathogenesis to vivo/vitro models and clinical therapeutic transformation. Int Wound J (2023) 20:4394–409. 10.1111/iwj.14311
20.
LenzenS. The mechanisms of alloxan- and streptozotocin-induced diabetes. Diabetologia (2008) 51:216–26. 10.1007/s00125-007-0886-7
21.
DeedsMCAndersonJMArmstrongASGastineauDAHiddingaHJJahangirAet alSingle dose streptozotocin-induced diabetes: considerations for study design in islet transplantation models. Lab Anim (2011) 45:131–40. 10.1258/la.2010.010090
22.
Matsumoto-OdaAUtsumiDTakahashiKHirataSNyachieoAChaiDet alInter-species differences in wound-healing rate: a comparative study involving primates and rodents. Proceedings R Soc B: Biol Sci (2025) 292 (2045): 20250233. 10.1098/rspb.2025.0233
23.
BenavidesFOberyszynTMVanBuskirkAMReeveVEKusewittDF. The hairless mouse in skin research. J Dermatol Sci (2009) 53:10–8. 10.1016/j.jdermsci.2008.08.012
24.
GurtnerGCWongVWSorkinMGlotzbachJPLongakerMT. Surgical approaches to create murine models of human wound healing. J Biomed Biotechnol (2011) 2011:969618. 10.1155/2011/969618
25.
Lopez-JornetPCamacho-AlonsoFGómez-GarciaFMolina MiñanoFCañasXSerafínAet alEffects of potassium apigenin and verbena extract on the wound healing process of SKH-1 mouse skin. Int Wound J (2014) 11(5):489–95. 10.1111/j.1742-481X.2012.01114.x
26.
BellRRDunstanRWKhanNK. Skin wound healing in the SKH-1 female mouse following inducible nitric oxide synthase inhibition. Br J Dermatol (2007) 157(4):656–61. 10.1111/j.1365-2133.2007.08096.x
27.
KoivunotkoEKoivuniemiRMonolaJHarjumäkiRPridgeonCSMadetojaMet alCellulase-assisted platelet-rich plasma release from nanofibrillated cellulose hydrogel enhances wound healing. J Controlled Release (2024) 368:397–412. 10.1016/j.jconrel.2024.02.041
28.
FurmanBL. Streptozotocin-induced diabetic models in mice and rats. Curr Protoc (2021) 1(4):e78. 10.1002/cpz1.78
29.
du SertNPAhluwaliaAAlamSAveyMTBakerMBrowneWJet alReporting animal research: explanation and elaboration for the arrive guidelines 2.0. PLoS Biol (2020) 18 (7): e3000411. 10.1371/journal.pbio.3000411
30.
MoreiraCCassini-VieiraPda SilvaMdaBL. Skin wound healing model - excisional wounding and assessment of lesion area. Bio Protoc (2015) 5 (22): e1661. 10.21769/bioprotoc.1661
31.
KoivuniemiRXuQSnirviJLara-SáezIMerivaaraALuukkoKet alComparison of the therapeutic effects of native and anionic nanofibrillar cellulose hydrogels for full-thickness skin wound healing. Micro (2021) 1(2):194–214. 10.3390/micro1020015
32.
BankheadPLoughreyMBFernándezJADombrowskiYMcArtDGDunnePDet alQuPath: open source software for digital pathology image analysis. Sci Rep (2017) 7(1):16878. 10.1038/s41598-017-17204-5
33.
SchafferBSGraysonMHWorthamJMKubicekCBMcCleishATPrajapatiSIet alImmune competency of a Hairless mouse strain for improved preclinical studies in genetically engineered mice. Mol Cancer Ther (2010) 9(8):2354–64. 10.1158/1535-7163.MCT-10-0207
34.
Silva-SantanaGBaxJCFernandesDCSBacellarDTLHooperCDiasAASOet alClinical hematological and biochemical parameters in Swiss, BALB/c, C57BL/6 and B6D2F1 Mus musculus. Anim Model Exp Med (2020) 3(4):304–15. 10.1002/ame2.12139
35.
SantosEWOliveiraDCdHastreiterASilvaGBdBeltranJSdeOet alHematological and biochemical reference values for C57BL/6, Swiss webster and BALB/c mice. Braz J Vet Res Anim Sci (2016) 53(2):138. 10.11606/issn.1678-4456.v53i2p138-145
36.
KolbH. Mouse models of insulin dependent diabetes: low‐dose streptozocin‐induced diabetes and nonobese diabetic (NOD) mice. Diabetes Metab Rev (1987) 3(3):751–78. 10.1002/dmr.5610030308
37.
KluehULiuZChoBOuyangTFeldmanBHenningTPet alContinuous glucose monitoring in normal mice and mice with prediabetes and diabetes. Diabetes Technol Ther (2006) 8(3):402–12. 10.1089/dia.2006.8.402
38.
LennikovAElZaridiFYangM. Modified streptozotocin-induced diabetic model in rodents. Anim Model Exp Med (2024) 7(5):777–80. 10.1002/ame2.12497
39.
ChaudhryZZMorrisDLMossDRSimsEKChiongYKonoTet alStreptozotocin is equally diabetogenic whether administered to fed or fasted mice. Lab Anim (2013) 47(4):257–65. 10.1177/0023677213489548
40.
AttrillEHScharapowOPereraSMayneSSumargoNRossRMet alControlled induction of type 2 diabetes in mice using high fat diet and osmotic-mini pump infused streptozotocin. Sci Rep (2025) 15(1):8812. 10.1038/s41598-025-89162-2
41.
CouturierACalissiCCracowskiJLSigaudo-RousselDKhouriCRoustitM. Mouse models of diabetes-related ulcers: a systematic review and network meta-analysis. EBioMedicine (2023) 98:104856. 10.1016/j.ebiom.2023.104856
42.
AnggraeniNSyamsunarnoMRAAWidyastutiRPuspitasariIMPraptamaS. Potential dual effect anti-inflammatory and anti-platelet of cogon grass ethanol extract on diabetic mice a preliminary study. J Phys Conf Ser (2019) 1246: 012006. 10.1088/1742-6596/1246/1/012006
43.
WylesSPDashtiPPirtskhalavaTTekinBInmanCGomezLSet alA chronic wound model to investigate skin cellular senescence. Aging. (2023) 15(8):2852–62. 10.18632/aging.204667
44.
RingEFJAmmerK. Infrared thermal imaging in medicine. Physiol Meas (2012) 33:R33–R46. 10.1088/0967-3334/33/3/R33
45.
Klama-BaryłaAKitalaDŁabuśWKrautMSzapskiMSmętekW. Infrared thermal imaging as a method of improving skin graft qualification procedure and skin graft survivability. Transpl Proc (2020) 52(7):2223–30. 10.1016/j.transproceed.2020.01.108
46.
FridbergMBaforAIobstCALaugesenBJepsenJFRahbekOet alThe role of thermography in assessment of wounds. A scoping review. Injury (2024) 55(11):111833. 10.1016/j.injury.2024.111833
47.
dos Santos-SilvaMATrajanoETLSchanuelFSMonte-Alto-CostaA. Heat delays skin wound healing in mice. Exp Biol Med (2017) 242 (3): 258–266. 10.1177/1535370216675066
48.
RattanSISFernandesRADemirovicDDymekBLimaCF. Heat stress and hormetin-induced hormesis in human cells: effects on aging, wound healing, angiogenesis, and differentiation. Dose-Response (2009) 7(1):90–103. 10.2203/dose-response.08-014.Rattan
49.
LiFXZLiuJJLeiLMLiYHXuFLinXet alMechanism of cold exposure delaying wound healing in mice. J Nanobiotechnology (2024) 22(1):723. 10.1186/s12951-024-03009-y
50.
WrightEHTylerMVojnovicBPleatJHarrisAFurnissD. Human model of burn injury that quantifies the benefit of cooling as a first aid measure. Br J Surg (2019) 106(11):1472–9. 10.1002/bjs.11263
51.
YangDRWangMYZhangCLWangY. Endothelial dysfunction in vascular complications of diabetes: a comprehensive review of mechanisms and implications. Front Endocrinol (2024) 15:1359255. 10.3389/fendo.2024.1359255
52.
ShaterianABorboaASawadaRCostantiniTPotenzaBCoimbraRet alReal-time analysis of the kinetics of angiogenesis and vascular permeability in an animal model of wound healing. Burns (2009) 35(6):811–7. 10.1016/j.burns.2008.12.012
53.
AitchesonSMFrentiuFDHurnSEEdwardsKMurrayRZ. Skin wound healing: normal macrophage function and macrophage dysfunction in diabetic wounds. Molecules (2021) 26:4917. 10.3390/molecules26164917
54.
AlSH. Macrophage phenotypes in normal and diabetic wound healing and therapeutic interventions. Cells (2022) 11 (15): 2430. 10.3390/cells11152430
55.
SasakiYOhsawaKKanazawaHKohsakaSImaiY. Iba1 is an actin-cross-linking protein in macrophages/microglia. Biochem Biophys Res Commun (2001) 286(2):292–7. 10.1006/bbrc.2001.5388
56.
KeshavSChungPMilonGGordonS. Lysozyme is an inducible marker of macrophage activation in murine tissues as demonstrated by in situ hybridization. J Exp Med (1991) 174(5):1049–58. 10.1084/jem.174.5.1049
57.
ZhaoRLiangHClarkeEJacksonCXueM. Inflammation in chronic wounds. Int J Mol Sci (2016) 17: 2085. 10.3390/ijms17122085
58.
SharmaSKishenA. Dysfunctional crosstalk between macrophages and fibroblasts under LPS-infected and hyperglycemic environment in diabetic wounds. Sci Rep (2025) 15(1):17233. 10.1038/s41598-025-00673-4
Summary
Keywords
collagen deposition, diabetogenic SKH1 mice, in vivo prolonged wound healing model, macrophage activity, STZ induction
Citation
Koivunotko E, Monola J, Pridgeon CS, Linden J, Harjumäki R, Yatkin E, Madetoja M and Yliperttula M (2026) Towards a standardized diabetic prolonged wound healing model in hairless SKH1 mice. Exp. Biol. Med. 251:10857. doi: 10.3389/ebm.2026.10857
Received
10 October 2025
Revised
21 December 2025
Accepted
16 February 2026
Published
20 March 2026
Volume
251 - 2026
Updates

Check for updates
Copyright
© 2026 Koivunotko, Monola, Pridgeon, Linden, Harjumäki, Yatkin, Madetoja and Yliperttula.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chris S. Pridgeon, christopher.pridgeon@helsinki.fi; Marjo Yliperttula, marjo.yliperttula@helsinki.fi
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.